 It also includes compliance to applicable regulatory requirements on a secondary basis. It is important to understand that the term risk, within the context of ISO 13485, applies primarily to the safety and performance of the medical device. What is the scope of these risks? How to determine the level of risk? The following questions, and the accompanying discussion, are intended to help you evaluate the need for additional improvement actions in your QMS. 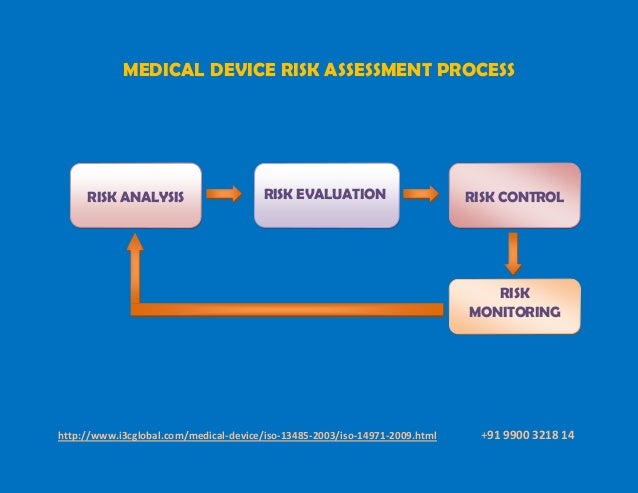
These newly added requirements can be difficult to interpret and implement. This high-level requirement then flows down to specific requirements under different clauses related to personnel, suppliers, verification of externally provided products/services and validation/re-validation of software. In simple terms, high risk processes are expected to have a higher level of control compared to lower risk processes. 
Control of these processes to achieve their desired results is now expected to be based on risk. It all starts with the role your organization (clause 4.1.1) plays in the lifecycle of the medical device, and the processes you have identified and implemented in your QMS (4.1.2(a)). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
